"..
Placing the elements into categories and subcategories based on shared properties is IMPERFECT. There is a spectrum of properties within each category and it is not hard to find overlaps at the boundaries, as is the case with most classification schemes.[23] Beryllium, for example, is classified as an alkaline earth metal although its amphoteric chemistry and tendency to mostly form covalent compounds are both attributes of a chemically weak or other metal. Radon is classified as a nonmetal and a noble gas yet has some cationic chemistry that is more characteristic of a metal.
Other classification schemes are Possible such as the division of the elements into mineralogical occurrence categories, or crystalline structures. Categorising the elements in this Fashion dates back to at least 1869 when Hinrichs[24] wrote that simple boundary lines could be drawn on the periodic table to show elements having like properties, such as the metals and the nonmetals, or the gaseous elements.
[........]
Other conventions and variations
[........]
Alternative layouts
Main article: Alternative periodic tables
Theodor Benfey's spiral periodic table
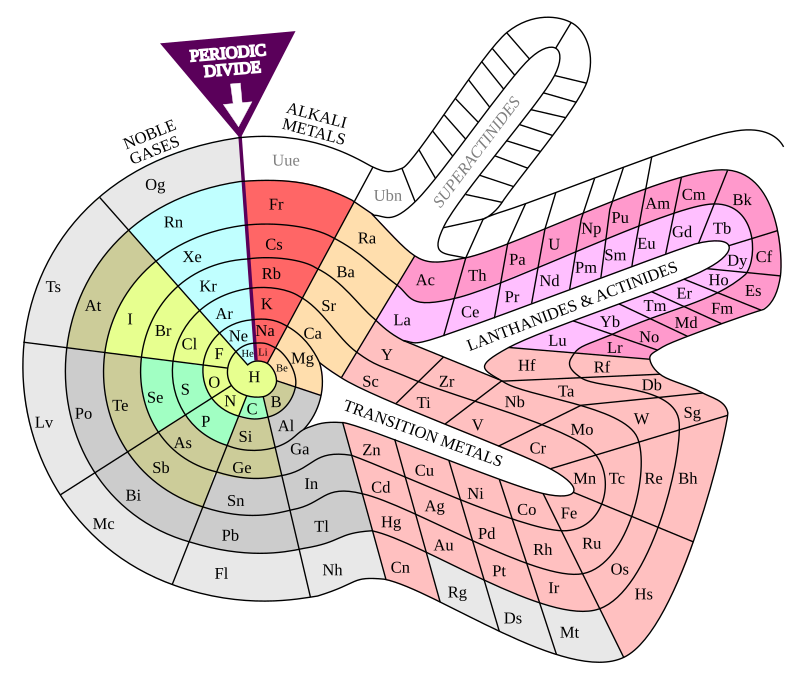
There are MANY periodic tables with layouts OTHER than that of the common or standard form. Within 100 years of the appearance of Mendeleev's table in 1869 it has been estimated that
around 700 Different periodic table versions were published.[81] As well as numerous rectangular variations, other periodic table formats have included, for example,[n 8] circular, cubic, cylindrical, edificial (building-like), helical, lemniscate, octagonal prismatic, pyramidal, separated, spherical, spiral, and triangular forms. Such alternatives are often developed to highlight or emphasize chemical or physical properties of the elements that are not as apparent in traditional periodic tables.[81]
A popular[82] alternative layout is that of Theodor Benfey (1960). The elements are arranged in a continuous spiral, with hydrogen at the center and the transition metals, lanthanides, and actinides occupying peninsulas.[83]
[.......]
Optimal form
The many different forms of periodic table have
prompted the Question of whether there is an optimal or definitive form of periodic table. The answer to this question is thought to depend on whether the chemical periodicity seen to occur among the elements has an underlying truth, effectively hard-wired into the universe, or if any such periodicity is instead the product of subjective human interpretation, contingent upon the circumstances, beliefs and predilections of human observers. An objective basis for chemical periodicity would settle the questions about the location of hydrogen and helium, and the composition of group 3. Such
an underlying truth, If it exists, is thought to have Not yet been discovered. In its absence, the many DIFFERENT forms of periodic table can be regarded as variations on the theme of chemical periodicity, each of which explores and emphasizes Different aspects, properties, perspectives and relationships of and among the elements.[n 12] The ubiquity of the standard or medium-long periodic table is thought to be a result of this layout having a good balance of features in terms of EASE of construction and size, and its depiction of atomic order and periodic trends.[50][121]""