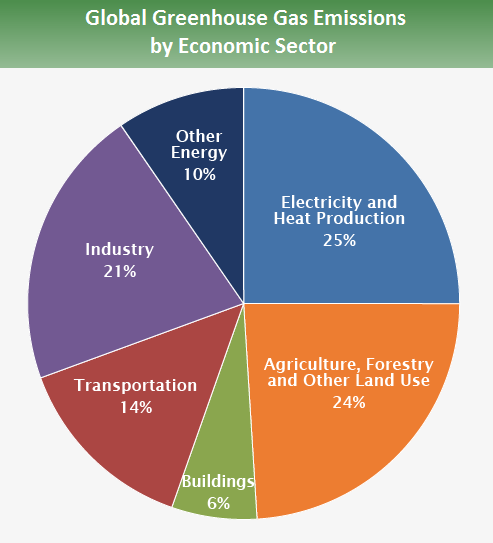
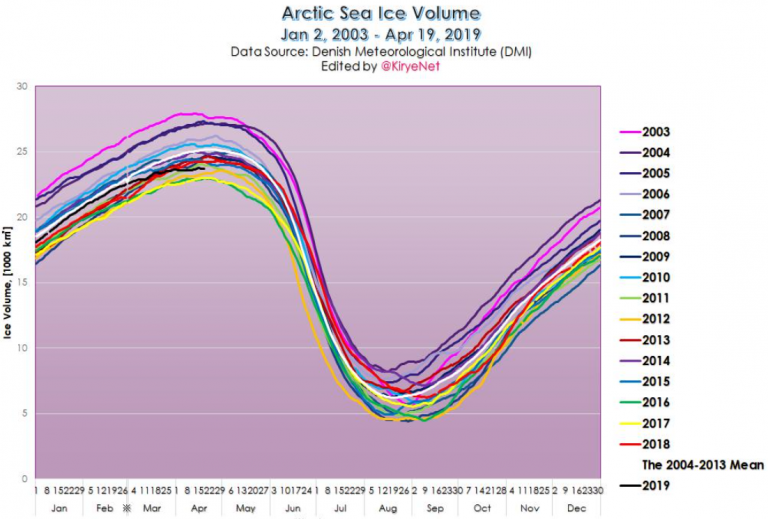
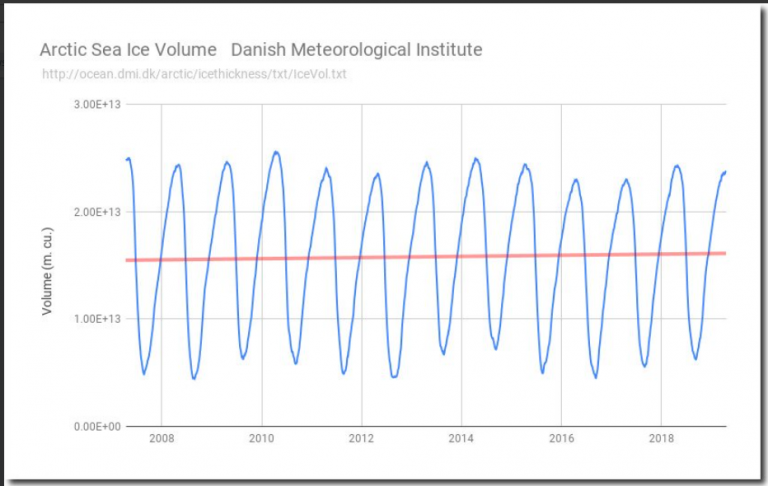
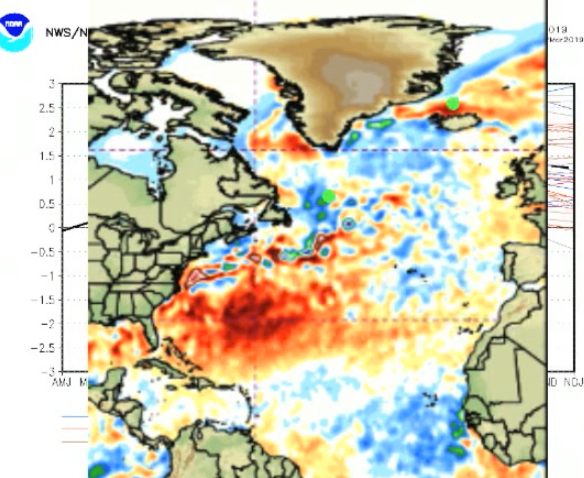
There you go, making ignorant assumptions again and proving you didn't read the link. Proving you don't understand science, and how variables are used in the formulas of physics.
One of the paragraphs:
(1) E = mc∆T
(2) E = mL
Where E is thermal energy (Joules), m is the mass (kg), c is the “specific heat” constant (J/kg/°C), ∆T is the change in temperature (°C), and L is the latent heat constant (J/kg). Specific heat is the amount of heat energy that we must add (or remove) from a specified mass to increase (or decrease) the temperature of that mass by 1 °C. Latent heat is the thermal energy released or absorbed during a constant temperature phase change. If we know the mass of the ice, water or atmosphere, it is easy to calculate the amount of energy it takes to change its temperature, melt it or freeze it.
Please note the use upper case "C" for Celsius, and lower case "c" for heat.
Have you ever done any practical science?